Research
Spontaneous Segregation of Hydrophilic Block Copolymers
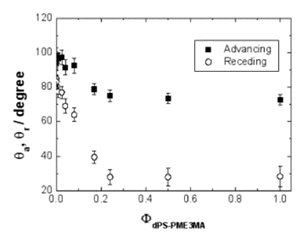
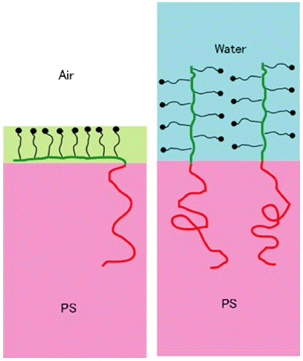
A block copolymer of deuterated polystyrene (dPS) and 2-[2-(2-methoxyethoxy)ethoxy]ethyl methacrylate (PME3MA) spontaneously exposes the PME3MA block, which is soluble in water, to the surface in a vacuum. dPS-PME3MA mixed with polystyrene (PS) segregates to the PS surface and changes the hydrophobic PS surface into hydrophilic surface. Neutron reflectivity, X-ray photoelectron spectroscopy and dynamic secondary ion mass spectrometry probe the surface segregation of dPS-PME3MA to the surface of PS due to its PME3MA block. Sum-frequency generation vibrational spectroscopy detects exclusively the triethylene oxide (3EO) side chains of PME3MA regardless of a small fraction of dPS-PME3MA in the mixture, suggesting the conformation of the side chains influencing the segregation behavior. Due to the amphiphilic character of the methoxy terminated 3EO side chains, the PME3MA block exposes the terminal methyl groups of the side chains to the surface to reduce the free energy of the system. However, such a hydrophobic methyl surface layer, which is only a few Å thick, barely covers the EO part of the side chains; the surface quickly reconstructs and becomes hydrophilic upon contact with water. This is a technologically important demonstration of converting a hydrophobic polymer surface to a hydrophilic (biologically inert) surface just by adding a small amount of a block copolymer.
The influence of ethylene oxide units in the side chain of hydrophilic block was also studied. Block copolymers of PS and hydrophilic blocks of 2-(2-methoxyethoxy)ethyl methacrylate (PS-PME2MA) and 2-methoxyethyl methacrylate (PS-PME1MA) showed the same segregation behaviour in air. However, the strength of the hydrophobic interaction in water of PME1MA modified surface is stronger than the other copolymers and the adsorption of protein was only observed on PME1MA surface. PME1MA is not water-soluble, whereas PME2MA and PME3MA are water-soluble. PS-PE2MA and PS-PME3MA block copolymers with water-soluble blocks are capable of forming brush layer in water, which prevent hydrophobic interaction and adsorption of protein.